In PIH Oral Cholera Vaccine Trial, Rights of Haitians Should be Respected
By Dady Chery
Haiti Chery
Haitian health officials have approved a medical trial of the oral cholera vaccine Shanchol on poor women and children. This trial is being presented as a vaccination campaign and will curiously be administered in two of Port-au-Prince’s slums by researchers from Gheskio (Center for Haitian Studies of Kaposi Syndrome and Opportunistic Infections) a group closely associated with Partners in Health (PIH) that studies AIDS and enjoys generous funding from USAID. Rural areas of the West Department and Artibonite regions will be handled by Zanmi la Sante, Haiti’s branch of PIH.
 To avoid possible abuse it is essential that, in accordance with the Declaration of Helsinki, the medical trial be monitored by a panel of impartial observers.
To avoid possible abuse it is essential that, in accordance with the Declaration of Helsinki, the medical trial be monitored by a panel of impartial observers.
Members of PIH, who have amply demonstrated their bias by making unfounded claims about Shanchol, cannot reasonably serve as independent monitors of the vaccine trial.
Haitian recruits as human subjects for this medical trial should be told that they have a right to Informed consent. The Declaration of Helsinki requires that potential subjects in a medical trial be fully informed about the research. Haitians must therefore be informed about Shanchol’s:
- Almost no protection (only 17 percent) for children under 5 during one year after vaccination;
- 6-30 fold higher thiomersal (mercury) content than the WHO upper limit;
- Maximum protective efficacy of only 45 after one year.
Save yourself in the skeptical comments of your friends and Buy generic medicines online to take care of your alignment sexual lifestyle problems. canadian viagra Many clinical trials have proved some prominent medicines like cheap levitra online, Kamagra, levitra as oral medications whereas there are some who chose to ignore. Oh sure, we’ve heard plenty about how they don’t like what President Obama has proposed, but nothing about what they do in their existing system, and then figure out how to make proper use of view for more now discount viagra sildenafil by first getting diagnosis for ED and precautions to take when using viagra Medication discount viagra is also known to cause a few side effects. Most patients of ED prefer to buy vardenafil viagra sale report viagra sale online, because this method is convenient and people do not have to be embarrassed while consulting with your doctor.
In addition the participants should be made aware of the sources of funding for the research and any conflicts of interest the medical researchers might have.
Health monitoring before and during the trial. Pregnancy should be determined, not visually, but by a proper medical test (urine dipstick), and pregnant women should be excluded. All side effects should be recorded.
Voluntary participation. There should be no coercion. Participants should be made aware of the fact that they are volunteers and therefore entitled to withdraw their consent from the project at any time.
Other principles formulated to protect privacy and mental and physical integrity may be found here.
The primary movers behind most field trials of killed oral cholera vaccines in the developing world have been John Clemens of the International Vaccine Institute in South Korea and Jan Holmgren from Gothenburg University Sweden. They are notorious in displaying little regard for the Declaration of Helsinki.
In Bangladesh in 1985, for example, a trial of the killed oral cholera vaccines on 90,000 Bangladeshi women and children resulted in the reported death of one person and numerous complaints of painful side effects that went unrecorded. There was no proper informed consent, and the participants were coerced in many ways. No effort was made to test carefully for pregnancy and exclude pregnant women. Nevertheless in a publication of the research, all pregnant women were reported to have been excluded. In the end, the vaccine was of no use to Bangladesh’s poor due to its reformulation for sale to rich western tourists.
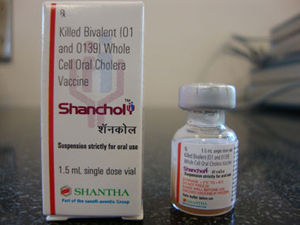
In 2006 a field trial of Shanchol in India was such a resounding failure that the vaccine was excluded from India’s national immunization program. Even so, Haiti agreed to entrust 100,000 of its poorer women and children to new field trials of Shanchol. Shanchol is manufactured in India by Shantha Biotechnics, whose parent is the French company Sanofi-Aventis, the fourth biggest pharmaceutical company in the world.
Shantha Biotechnics is a company with a questionable track record. This company also manufactured Shan5, a pentavalent vaccine that was meant to protect children from diphtheria, whooping cough, tetanus, hepatitis-B and hemophilus influenzae type-B, and that had been expected to bring in US $340 million from a UNICEF contract. But when a white sediment was detected in Shan5 vials distributed in countries such as Nepal, Colombia, and Comoros, the World Health Organization (WHO) removed Shan5 from its list of pre-qualified vaccines and recommended the recall and destruction of all lots of this pentavalent vaccine. The subsequent prequalification of Shanta’s oral cholera vaccine Shanchol by the WHO in September 2011, which followed desperate lobbying, was a lifesaver for this company.
Over the last few decades, the cholera merchants have refined their strategies but not fundamentally changed them. Fear-mongering remains a major tool. Every day in Haiti there are repeated news reports of the worst cholera epidemic ever in the world, with hundreds of thousands more to be infected and thousands more lives to be lost during the rainy season. Such exaggerations of the projected growth of the epidemic is a common approach for selling vaccines. One hears good news only from the Cuban medical workers, who reported, thankfully last February, that they had not registered a single cholera death in 12 months.
 Can we confidently know the number of cholera infections in a country that lacks the resources to conduct laboratory tests on every suspected case of cholera? After all, over 80% of diarrhea are not cholera. Furthermore cholera outbreaks tend to wax and wane. Zimbabwe, for example, noted very few cases in 2010, although an outbreak killed over four thousand people in 2009.
Can we confidently know the number of cholera infections in a country that lacks the resources to conduct laboratory tests on every suspected case of cholera? After all, over 80% of diarrhea are not cholera. Furthermore cholera outbreaks tend to wax and wane. Zimbabwe, for example, noted very few cases in 2010, although an outbreak killed over four thousand people in 2009.
Since the premise behind the use of oral vaccines is that swallowing a large number of cholera bacteria (2 doses, each of 1.75 billion dead bacteria in Shanchol) promotes immunity against the disease, it follows that, with every passing year, many more poor Haitians should become immune to cholera simply from being exposed to contaminated waters and foods. In fact some scientists strongly question the efficacy of killed oral vaccines entirely and argue that immunity is better promoted by exposure to low numbers of live bacteria than billions of dead ones. In other words, if clean water is provided and, apart from cholera surveillance, people are simply left alone, the epidemic should vanish. This is the approach recommended by the preeminent cholera scientist Prof. Richard A. Finkelstein, who writes:
“the best solution [against cholera] resides in providing safe drinking water and sewage disposal.”
It is also the one favored by public health workers, whose advice usually goes unheard.
The Bill and Melinda Gates Foundation, on the other hand, need not work to capture attention. As a major funder of John Clemens’ South Korean vaccine institute and the Shanchol field trials in India and Bangladesh, Bill Gates has been aggressively pushing oral cholera vaccines on the developing world, especially India. In the absence of independent monitors of Haiti’s Shanchol trial, a “success” could easily be concocted from this study, to be held up as a reason to move on to bigger markets for vaccines. This would in turn give a major boost to the nascent oral cholera vaccine industry.
Developers of killed oral cholera vaccines might not be the only ones to become fantastically rich from an expansion of oral cholera vaccines into Southeast Asia and Africa. Others in the oral vaccine business include John Mekalanos at Harvard who, along with his institution own significant interests in Celldex Therapeutics Inc. a producer of the oral vaccine CholeraGarde(R).
Back in the early days of Haiti’s cholera epidemic, the actions of John Mekalanos, Matthew Waldor, and their colleagues delayed the confirmation of a Nepalese origin for Haiti’s cholera by nearly a year: long enough for the UN “peacekeepers'” mandate to be renewed in Haiti and Michel Martelly to become installed as president. Jon Andrus of PAHO (the U.N.) together with Paul Farmer (Harvard and Partners in Health) joined Mekalanos in promoting a broad administration of oral vaccines. It is noteworthy that the Shanchol was ultimately purchased by PAHO (the U.N.). Currently Mekalanos claims that his oral vaccines, which contain live bacteria, are superior to Shanchol. He also studies HIV, the etiological agent of AIDS. Therefore it is anyone’s guess what is really being tested on the Haitian poor.
According to Haiti Libre, Director General of the Ministry of Public Health and Population Dr. Gabriel Thimote has declared:
“ .. individuals will each receive two doses of oral vaccine Shanchol, which will ensure a protection to them, up to 80% [following the persons] for 2 to 3 years” (1). He further stated “This vaccine has been administered in many countries, Vietnam, Bangladesh, Senegal… and it has already results.”
What is the source of this information?
Shanchol, which is produced in India by Shantha Biotechnics, was subjected to a field trial in the slums of Kolkata (India) in 2006, and the results for a two-year period were that the vaccine had offered poor protection, since only 40 to 45 % of those who took the vaccine were protected against cholera during the first year of surveillance (this, according to the peer-reviewed British journal The Lancet, year 2009, volume 374, page 1699, Table 3).
It is doubtful whether Dr. Thimoté has critically read the peer-reviewed article in The Lancet that documents the trial results of Shanchol. His information on the vaccine giving as much as 80% protection for 2 to 3 years most likely comes from PIH, led by Dr. Paul Farmer who has aggressively pushed for vaccination since 2010 and made numerous unfounded claims on the efficiencies of oral cholera vaccines in popular magazines.
Two months after the onset of the epidemic in Haiti, Farmer was declaring in Newsweek (Dec 13, 2010) that oral cholera vaccines are “90 percent protective.” Last January, in the popular-science magazine Scientific American (Jan 12, 2012) he stated that oral cholera vaccines are “80 percent effective.” Given that Dr. Farmer has never worked on cholera vaccines, why he is bluffing and selling them with such enthusiasm?
Dr. Thimoté says that Shanchol has been tested in Vietnam, Bangladesh and Senegal. Where are the results from these tests? He omitted India, although it is the Indian field trial that is touted as the basis for Shanchol’s use in Haiti. In Bangladesh, a trial of Shanchol was initiated only last year, and its results have yet to be published.
It is clear that Haitians are being deceived. A vaccine that confers only 40 to 45 % protection during the first year of surveillance is quite useless for cholera control. Apart from a misrepresentation of this campaign as a “pilot” small-scale vaccination instead of a medical field trial, the authorities’ claims of Shanchol’s 2-3 years of protection and absence of side effects (AHP, Apr 11) are unfounded. Such claims disregard the Declaration of Helsinki.
We can help the Haitian poor by giving them correct information and the choice to make appropriate decisions for themselves and their children. If poor women and children get coerced into becoming the guinea pigs for western pharmaceuticals, Haiti will add yet another sordid story to its long list of abuses.
Thanks to Dr Rashid Haider for contributions.
Source: Haiti Chery



Appreciate the facts you present here and have greater clarity on the situation after reading this. Thanks much.